
From Durham with love, Mahlkoenig staff Kyle Ramage and his frozen coffee beans became the nova of 36 competitors in the United States Barista Championship(USBC) 2017 and wonderfully won the trophy. He, and his dry ice frozen coffee has become the most eye-catching part in the competition-frozen to-79℃, then grind and brew.
He mentioned Dr.Christopher H. Hendon (Chris in following), his tech consultant, co-author of Water for coffee in 2015 and “The effect of bean origin and temperature on grinding roasted coffee” (Uman, E., Colonna-Dashwood, M., Colonna-Dashwood, L., Perger, M., Klatt, C., Leighton, S., Miller, B., Butler, K. T., Melot, B. C., Speirs, R. W., Hendon, C.H., Sci. Rep., 2016, 6, 24483) in 2016. How ever, nominating Dr. Christopher, H.Hendon’s achievements with “2015 Water for Coffee” and “2016 Freezing beans”, as he says, is a particularly narrow summary. First of all, his ‘achievements’ should start with obtaining a PhD in Chemistry at the University of Bath, as well as doing a Postdoc at Massachusetts Institute of Technology. He is now an Assistant Professor at the University of Oregon. Of his 40+ peer-reviewed publications, three involve coffee, and a book “Water for Coffee”. In 2016, he gave a presentation on the many quantitative data and instruments used in the preparation of coffee, challenging many “certainties” that often interfere with a true qualitative approach.
Then, he started to ground some cold coffee.In his research with other authors, it has been revealed that ‘Grinding colder coffee beans produces a more uniform particle distribution, with a decreased particle size. While the decreased particle size will tend to speed up extraction due to the larger surface area, the increased uniformity should minimise the amount of wasted bean, which is discarded without being extracted to completion.’
CTI=Coffee t&i
Chris=Dr. Christopher H. Hendon

CTI: How you look at this comment? Do you agree?
“At a coffee shop, freezing beans is especially impractical. Baristas cope with the morning rush by having a hopper full of beans ready to grind at the flip of a switch. Making coffee from individually frozen packets of beans would take too long. In this sense, the current study may be tantalizing as much as anything else — after all, lots of folks need coffee to wake up, but it’s only going to be the early risers who get the best cups.”
Chris: Freezing coffee is certainly more challenging than a typical workflow in a cafe… but the benefits might outweigh the cost of training the staff to efficiently work with a single dose grinder and a freezer. If you are a typical café with high volume peaks throughout the day, you might actually find the freezing allows you to achieve higher quality consistently, because you don’t have those ‘hot’ periods as the grinder cools.
I don’t think that the study is intended to change the way we make coffee in cafes. In fact, quite the opposite. I think that the study explains and clearly shows that temperature affects the physics of the grinder. Whether you then go and start monitoring burr and bean temperatures is up to you, but it should have emphasized the importance of this knowledge.
I do believe, however, that the ‘early riser gets the best cup’ is a terrible business model. Because this is simply not true. Once the grinder cools after the rush, the coffee should be the same as before the rush. It is just the period during high volume grinding.
CTI: Any advice for distinguishing relevant and qualitative data and instruments?
Chris: Sure. You need to first determine what you are trying to measure and why. For example, I might want to measure how much coffee by mass I have extracted. But why? The answer I will leave for you to discuss, but I can tell you that the ‘why’ to this question does not provide insight towards our end goal: tasty coffee.
The most valuable method for distinguishing if a device is proving valuable data is if it is reporting in SI units. For example if you want to measure a flow rate the device would need to report in units of volume per time (e.g. 3 mL/s). A device that reports in some arbitrary unit is not flawed, but does require further inquisition. I trust thermometers, pressure gauges and time.

CTI: Did you collaborate with Kyle, expand your findings to specific beans and grind level?
Chris: I have known Kyle for many years. He contacted me to tell me he wanted to use this cold coffee idea in his routine. I was excited, I like competition. He asked for the same graph that we published, but a little bit less complex (we removed the freezer and liquid nitrogen temperature traces). Note that in the Sci. Rep. paper, we showed that all beans ground the same, independent of the origin, processing method, roast or roaster… so we didn’t need to expand the study beyond our as reported data. I just tell you what happens relative to room temperature, and you – the barista – then has to work out how to make it taste good.
CTI: Which point was the most challenging and how your overcame it?
Chris: Nothing is particularly challenging. Kyle is extremely smart and was a great student. The key was for him to get his coffees frozen, ground frozen, and then tasting great. He had Lem Butler to do that with, so that was easy!
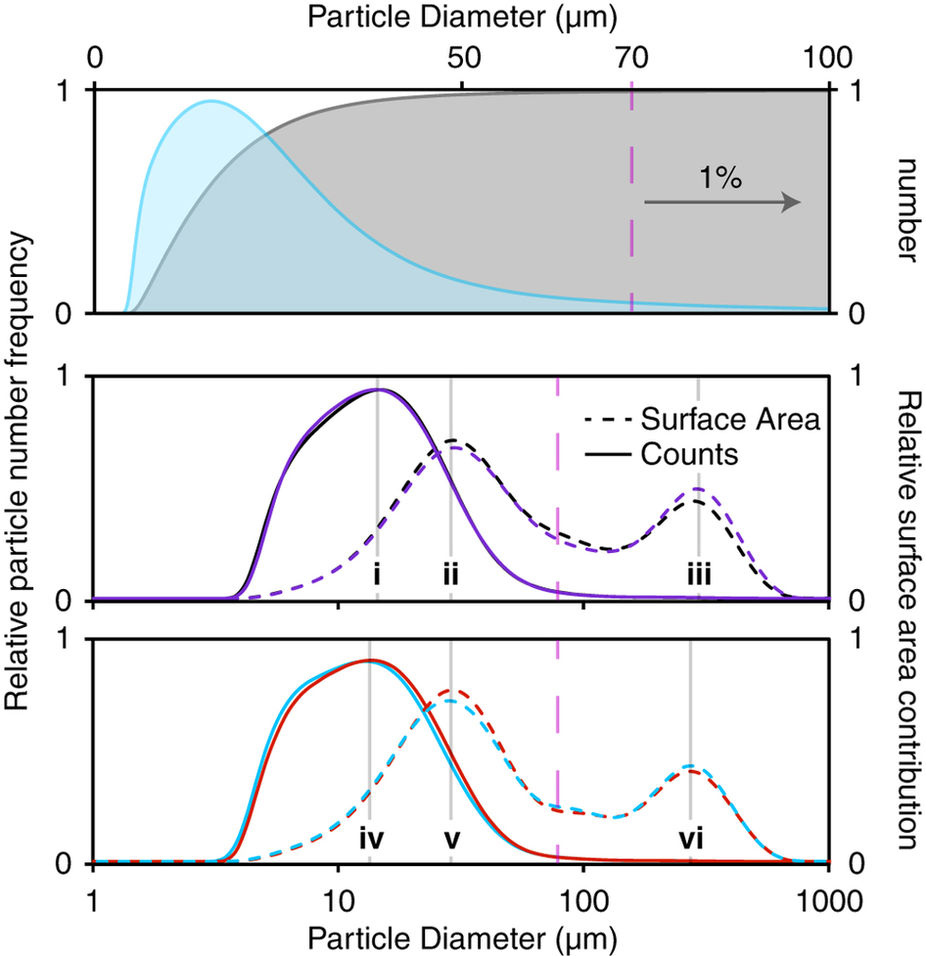
CTI: How to implement into everyday productions? Will there be any course?
Chris: If you vacuum seal your coffee doses you can freeze them forever. Whatever you want to do with that is up to you. Sometimes its neat to freeze it and then keep it for a year to compare to the next crop. The applications are endless. I don’t think I will run a course, because the concept is simple!
CTI: Or, still-“The question was always how does temperature affect particle size in grinding. What you conclude from the data is up to you”.
Chris: This is still true.
CTI: What are your motivates contributing to coffee industry nowadays?
Chris: How simple chemical concepts like polarity or kinetics can be observed in real life. And, at this point, I realized coffee is an excellent medium for teaching chemistry. Everyone likes chemistry and coffee. Its just that the latter is much easier to get into. But honestly, I think the world of coffee has so many untapped curiosities and I am happy to explore them.

CTI: Would you like to talk about “Water for Coffee” and those papers involving coffee?
Chris: The first paper, written with Maxwell and Lesley Colonna-Dashwood detailed why we like Mg2+ and Ca2+ in our water. Following on from the came concepts of interactions of coffee molecules with water the second paper details a very complex analysis of why sugar addition to coffee is likely to change nothing chemically. Rather it probably just changes how we perceive the flavors in coffee (humans like sugar and are sensitive to it).
Water For Coffee contains these concepts, and more, and was intended to teach the community why the chemistry matters. It didn’t offer too many solutions to problems, leaving ample room for innovations like Third Wave Water, and other products that attempt to standardize the water chemistry.
CTI: What’s your plan next?
Chris: I am at the junction of postdoctoral research and launching my own research group. To date, the coffee industry has been fruitful for both my career and my abilities as a communicator and teacher. I am moving to Eugene, Oregon to be a professor in Chemistry. This will happen the end of June.
CTI: Can we find your journals in different languages in the future?
Chris: Peer-reviewed articles are never translated in science, sorry! But, there is a plan to sell Water For Coffee to a Chinese publisher for translation and legal production in China.
Dr. Christopher H. Hendon’s papers on coffee:
“The role of dissolved cations in coffee extraction”
Hendon, C. H., Colonna-Dashwood, L., Colonna-Dashwood, M., J. Agric. Food Chem., 2014, 62, 4947
“The impact of solvent relative permittivity on the dimerization of organic molecules well below their solubility limits: examples from brewed coffee and beyond”
Bradley, E. S., Hendon, C. H., Food Funct., 2017, 8, 1037
“The effect of bean origin and temperature on grinding roasted coffee”
Uman, E., Colonna-Dashwood, M., Colonna-Dashwood, L., Perger, M., Klatt, C., Leighton, S., Miller, B., Butler, K. T., Melot, B. C., Speirs, R. W., Hendon, C.H., Sci. Rep., 2016, 6, 24483









NO COMMENT